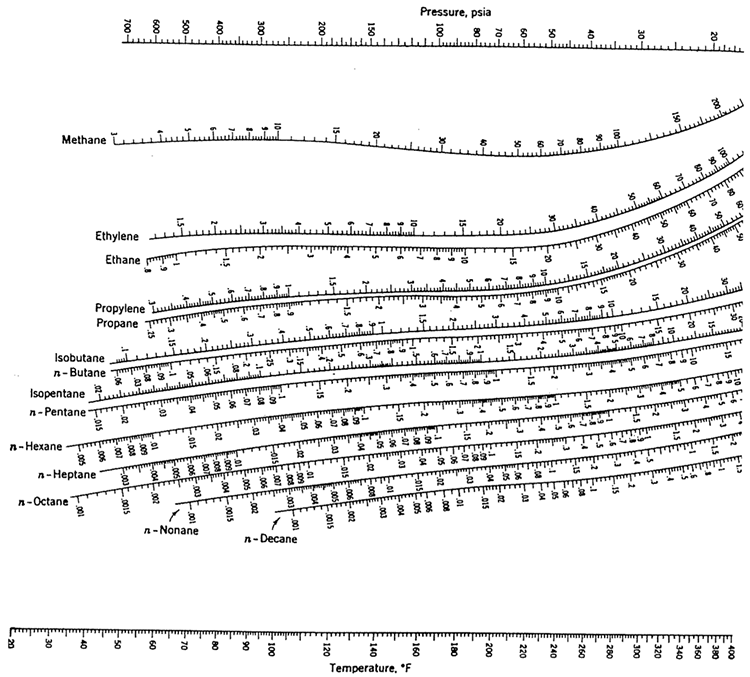
Note where the line crosses the methane axis.Connect the points with a straight line.On the right-hand vertical axis, locate and mark the point containing the temperature 60☏.DePriester in an article in Chemical Engineering Progress in 1953.
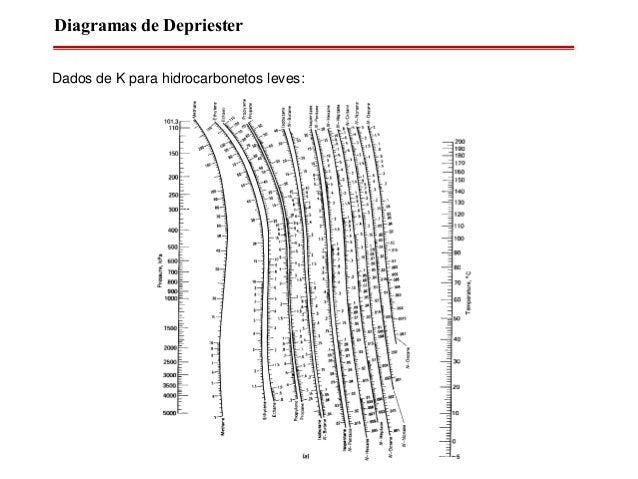
On the left-hand vertical axis, locate and mark the point containing the pressure 100 psia. DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature.Example įor example, to find the K value of methane at 100 psia and 60 ☏. Many DePriester charts have been printed for simple hydrocarbons. Based on Antoine equation, the boiling point for pure D is given by. yn+1 1 R R+ xn + 1 R+1 xD Let slope 1 R R+ and yint 1 R+1 xD then. ( These will be used to generate initial guesses later on ). When the bubble point calculation using the liquid mole fraction from the stripping operating line produces the vapor mole fraction of n-pentane exceeding yq, the liquid mole fraction of pentane entering an equilibrium stage is determined from the rectifying operating line. Solve for the boiling points of pure compounds. the pressure, the Antoine coefficients, etc. A chart can be obtained by going to the following link and downloading. Using DePriester charts, estimate the bubble point temperature of a mixture of 25(by mol) n-petane, 45 n-hexane, and 30 n-heptane mixture at 101.3 kpa. "K" values, representing the tendency of a given chemical species to partition itself preferentially between liquid and vapor phases, are plotted in between. Setup the spreadsheet with the necessary parameters, e.g. Note that for doing the following problem, DePriester Charts are required. Using DePriester charts, estimate the bubble point temperature of a mixture of 25(by mol) n-petane, 45 n-hexane, and 30 n-heptane mixture at 101.3 kpa. These nomograms have two vertical coordinates, one for pressure, and another for temperature. DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature.
